Quinolones 1. Older (Earlier) quinolones include: Nalidixic acid, oxalinic acid and cinoxacin. 2. Fluorinated derivatives (Fluoroquinolones) include : - ppt download
Resurrection of Nalidixic Acid: Evaluation of Water-Based Nanoformulations as Potential Nanomedicine | Nanoscale Research Letters | Full Text
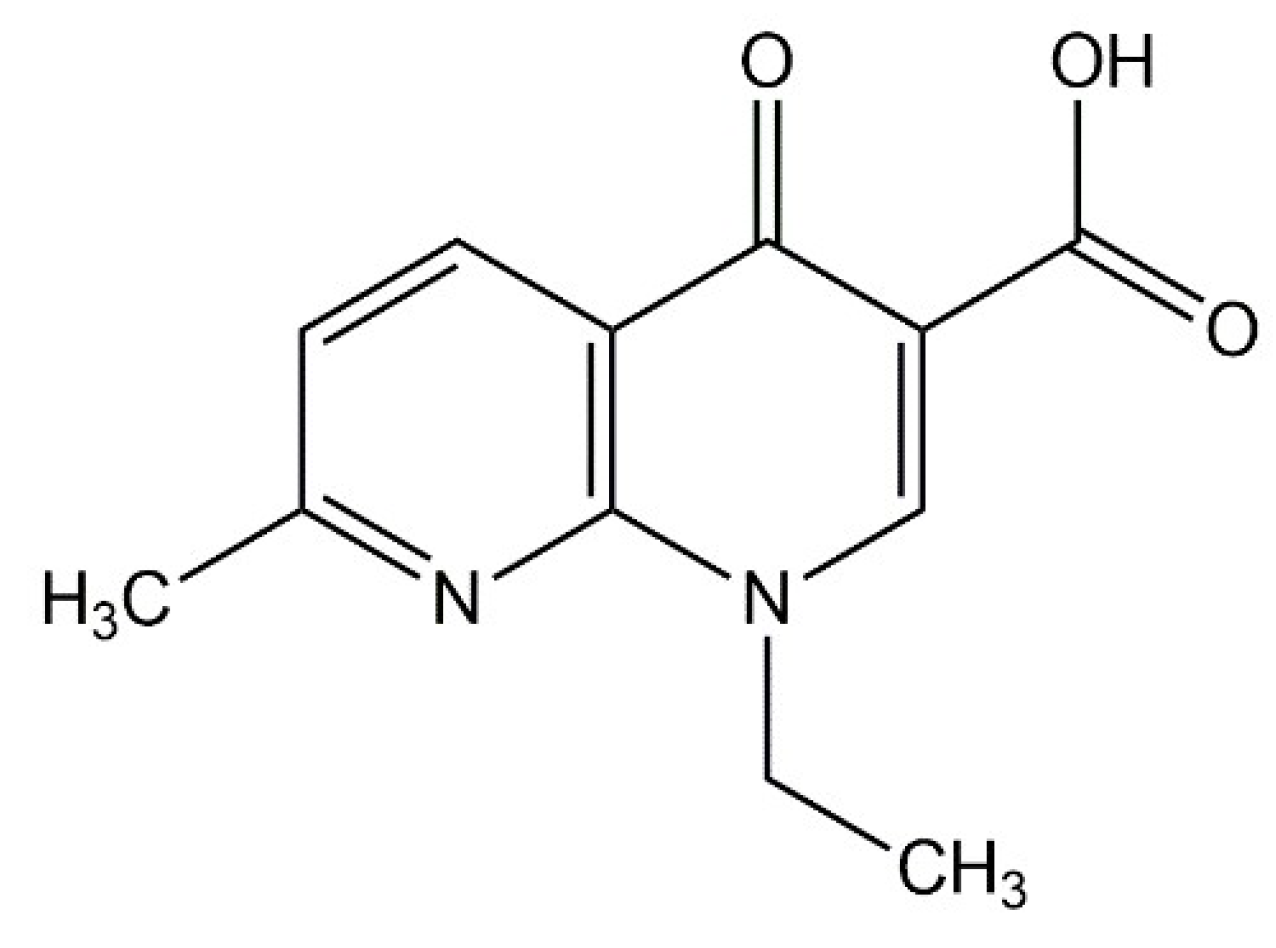
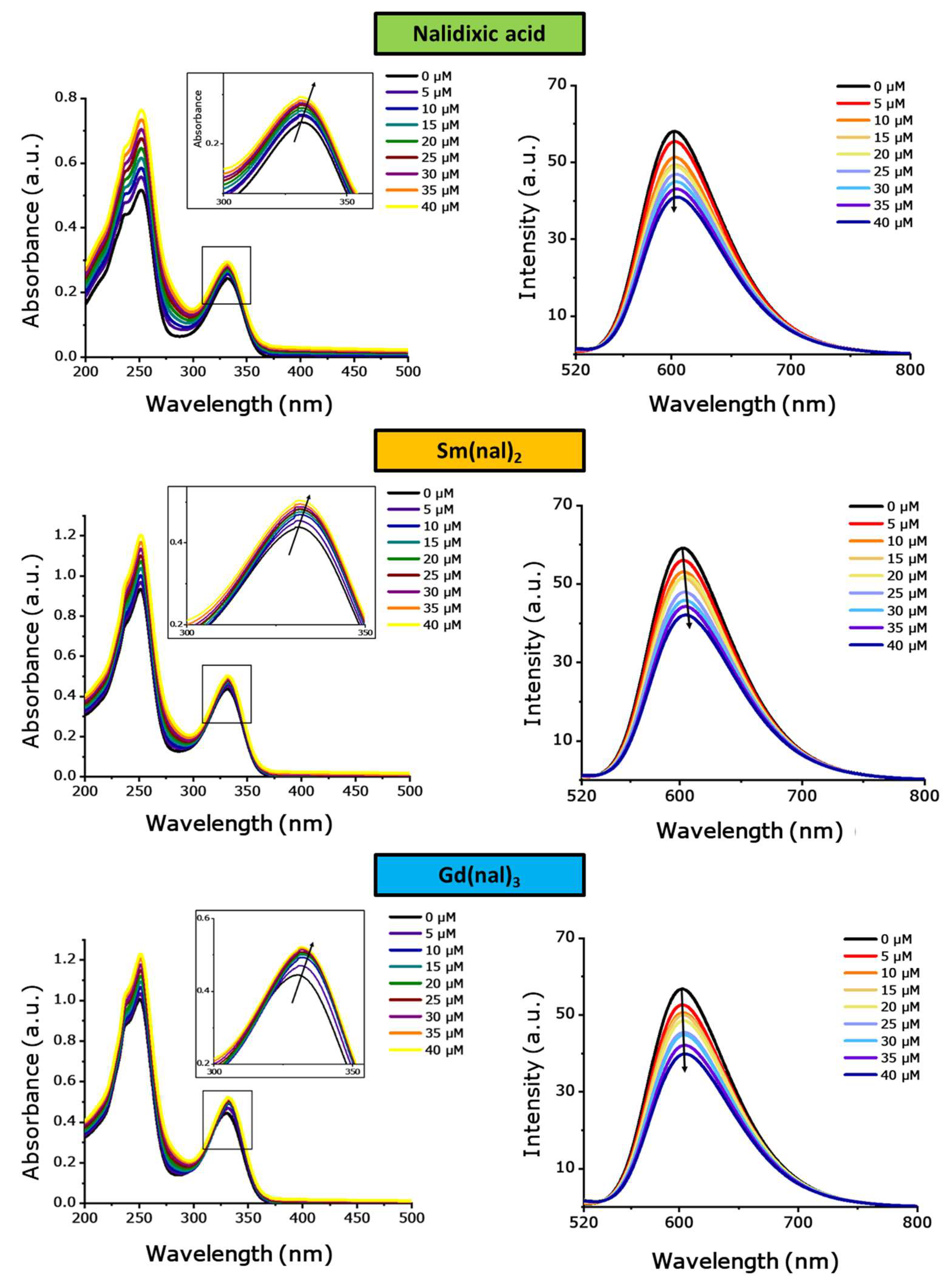
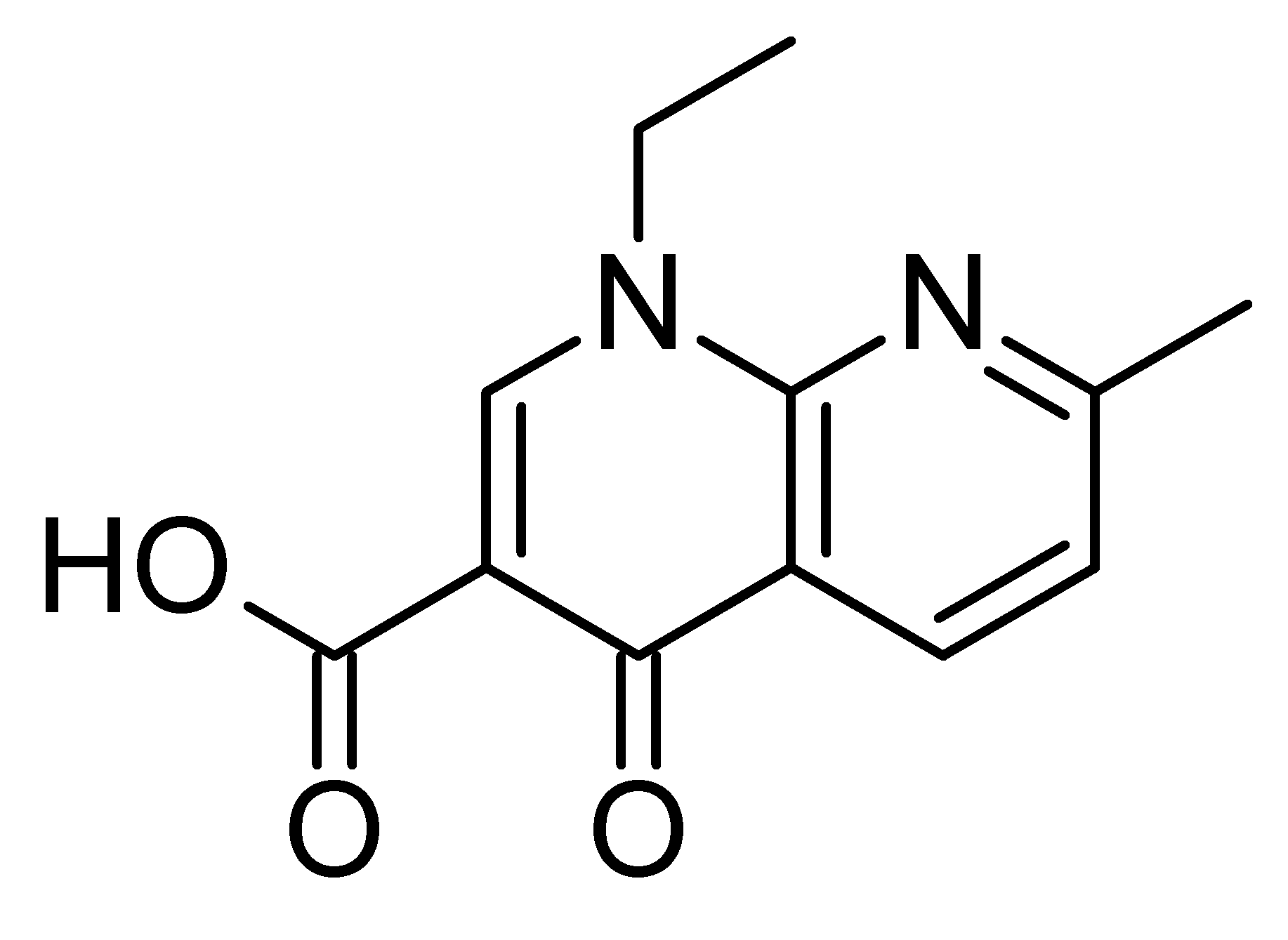
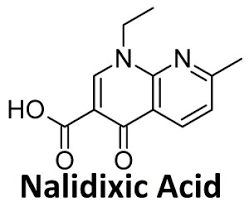
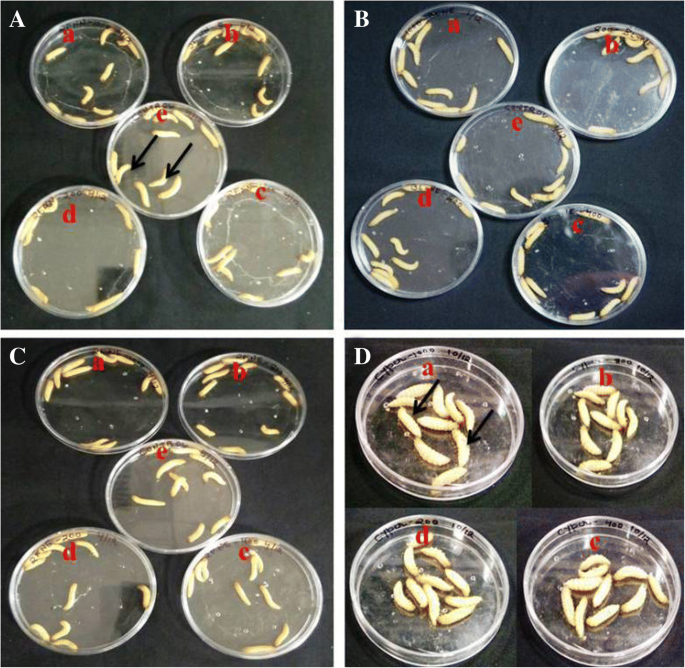
Pharmaceuticals | Free Full-Text | A Study on Repositioning Nalidixic Acid via Lanthanide Complexation: Synthesis, Characterization, Cytotoxicity and DNA/Protein Binding Studies | HTML
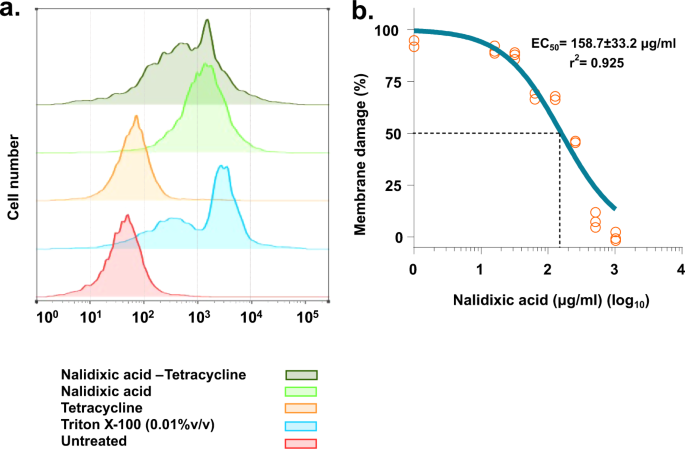
Mechanistic insights into synergy between nalidixic acid and tetracycline against clinical isolates of Acinetobacter baumannii and Escherichia coli | Communications Biology
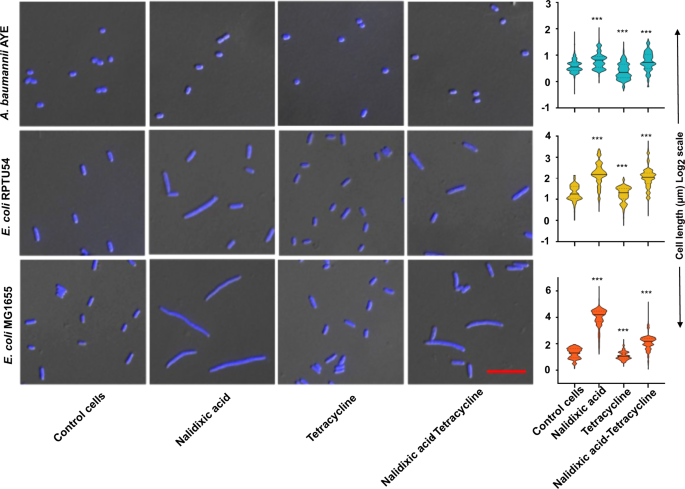
Mechanistic insights into synergy between nalidixic acid and tetracycline against clinical isolates of Acinetobacter baumannii and Escherichia coli | Communications Biology
Use of a MAMA-PCR Method to detect gyrA Mutations in Nalidixic Acid Resistant Clinical Isolates of Escherichia coli
The effect of infinitesimal drug dilutions on the pharmacokinetics of nalidixic acid and atenolol. - Abstract - Europe PMC