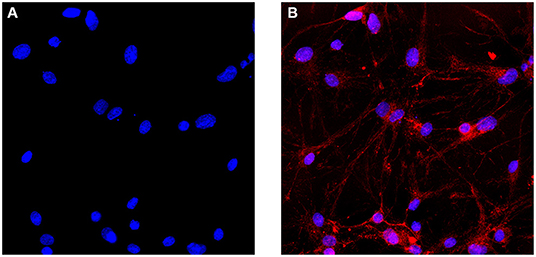
Frontiers | Ozone Exposure Induces Metabolic Disorders and NAD+ Depletion Through PARP1 Activation in Spinal Cord Neurons

Scope and Limitations of Typical Copper-Free Bioorthogonal Reactions with DNA: Reactive 2′-Deoxyuridine Triphosphates for Postsynthetic Labeling | The Journal of Organic Chemistry
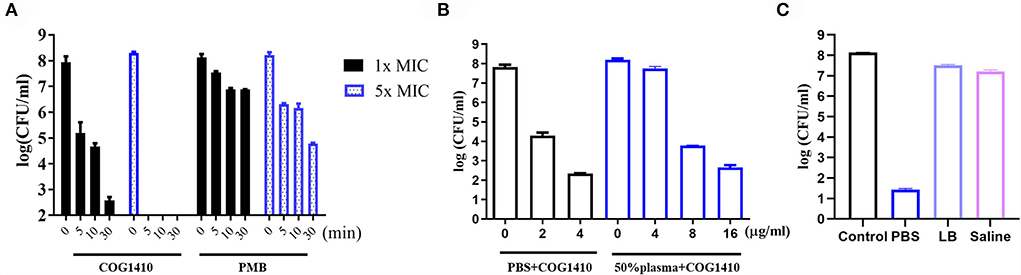
Frontiers | Apolipoprotein E mimetic peptide COG1410 combats pandrug-resistant Acinetobacter baumannii

Enhanced Activity against Multidrug-Resistant Bacteria through Coapplication of an Analogue of Tachyplesin I and an Inhibitor of the QseC/B Signaling Pathway | Journal of Medicinal Chemistry

Satellite-Based Land-Use Regression for Continental-Scale Long-Term Ambient PM2.5 Exposure Assessment in Australia | Environmental Science & Technology

Synthesis, Characterization, and Cytotoxicity of Morpholine-Containing Ruthenium(II) p-Cymene Complexes | Inorganic Chemistry

Combining Chemical Genetics with Proximity-Dependent Labeling Reveals Cellular Targets of Poly(ADP-ribose) Polymerase 14 (PARP14) | ACS Chemical Biology
Evaluation of antibodies to oxidized low-density lipoprotein and assessment of C-reactive protein in acute coronary syndrome and

Phenol Derivatives as Coupling Partners with Alkylsilicates in Photoredox/Nickel Dual Catalysis | The Journal of Organic Chemistry

Chemical and Structural Variety in Sodium Thioarsenate Glasses Studied by Neutron Diffraction and Supported by First-Principles Simulations | Inorganic Chemistry

Studying Excipient Modulated Physical Stability and Viscosity of Monoclonal Antibody Formulations Using Small-Angle Scattering | Molecular Pharmaceutics

Chemoenzymatic Synthesis of a Library of Human Milk Oligosaccharides | The Journal of Organic Chemistry

Redox Properties of Ruthenium Nitrosyl Porphyrin Complexes with Different Axial Ligation: Structural, Spectroelectrochemical (IR, UV−Visible, and EPR), and Theoretical Studies | Inorganic Chemistry

ACS Academica Transilvania - ASA Tirgu-Mures | ACS Academica Transilvania - ASA Tirgu-Mures | By ACS Academica Transilvania Tîrgu-Mureș | Facebook
![PDF) Highly Antiproliferative Latonduine and Indolo[2,3-c]quinoline Derivatives: Complex Formation with Copper(II) Markedly Changes the Kinase Inhibitory Profile PDF) Highly Antiproliferative Latonduine and Indolo[2,3-c]quinoline Derivatives: Complex Formation with Copper(II) Markedly Changes the Kinase Inhibitory Profile](https://i1.rgstatic.net/publication/358321290_Highly_Antiproliferative_Latonduine_and_Indolo23-cquinoline_Derivatives_Complex_Formation_with_CopperII_Markedly_Changes_the_Kinase_Inhibitory_Profile/links/620e7a5ff02286737ca5f76d/largepreview.png)