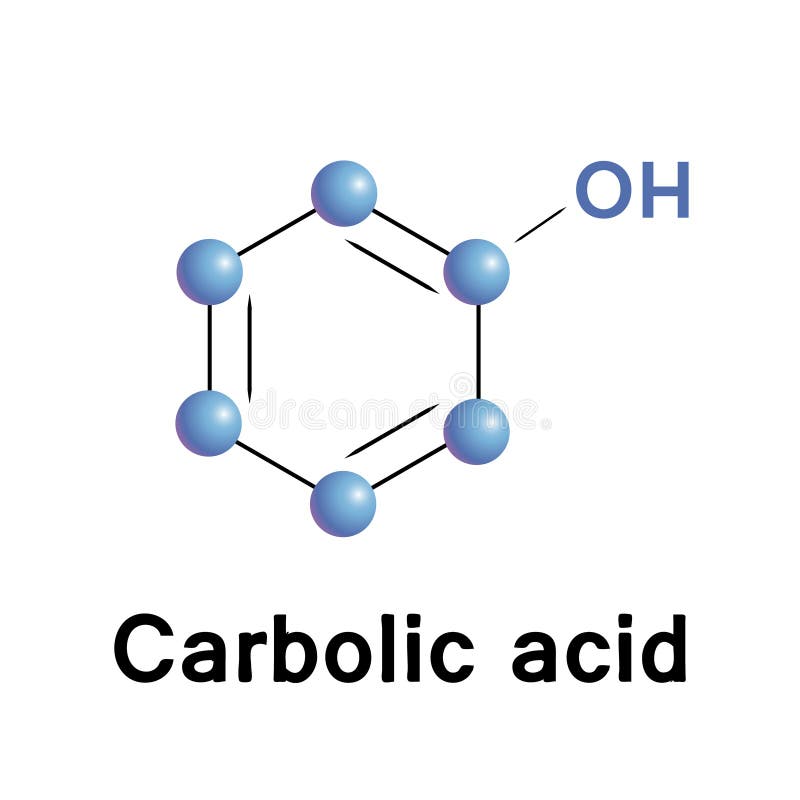
Phenol (carbolic acid) — is an aromatic organic compound with the molecular formula C6H5OH. It is a white crystalline solid that is volatile Stock Photo - Alamy

Atomic Chemical Structure Formula Phenol Molecular Stock Vector (Royalty Free) 2041189733 | Shutterstock
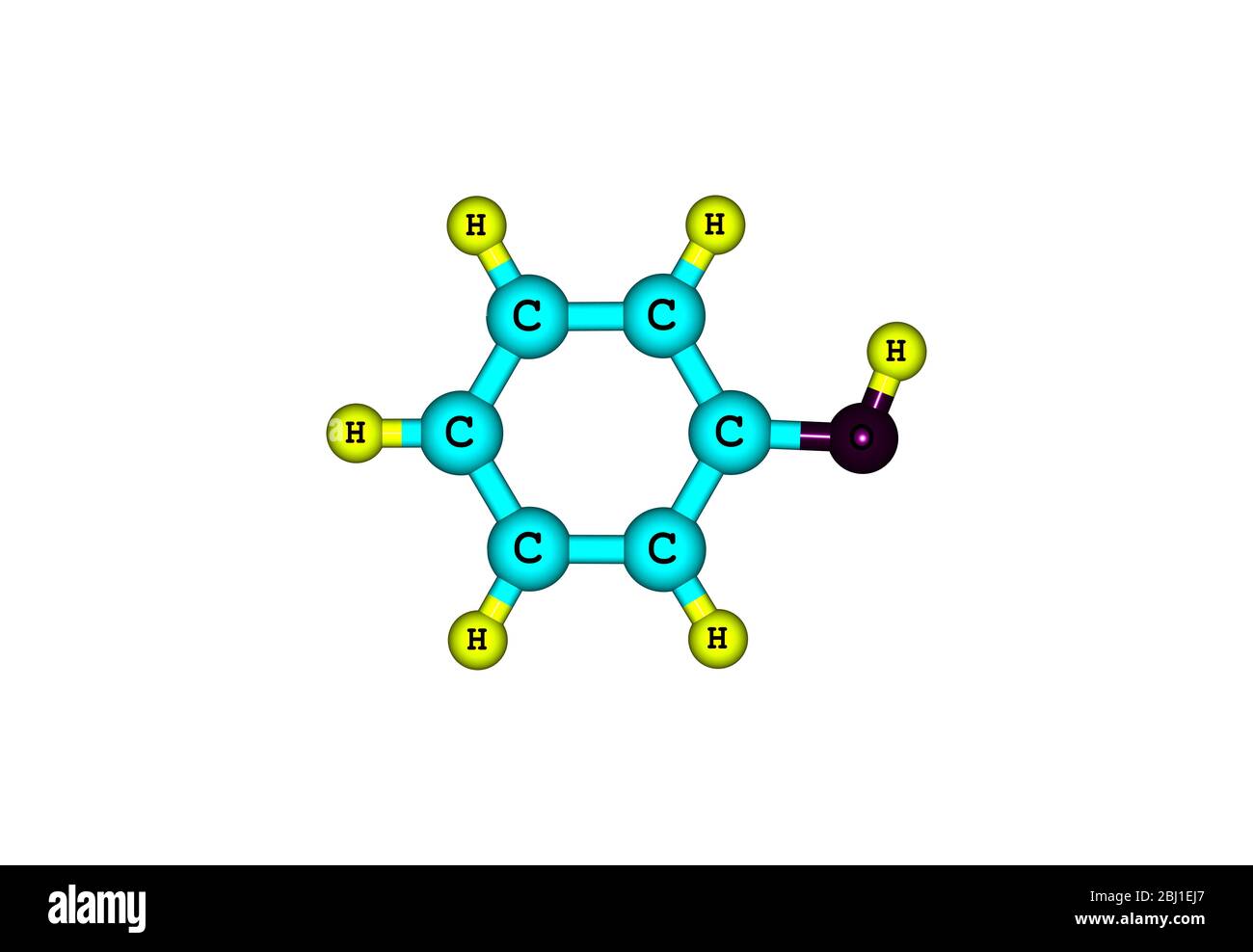
Vector Ballandstick Model Of Chemical Alcohol Icon Of Phenol Molecule C6h5oh Consisting Of Carbon Oxygen And Hydrogen Structural Formula Suitable For Education Isolated On A White Background Stock Illustration - Download Image

1H proton nmr spectrum of phenol C6H6O C6H5OH low/high resolution analysis interpretation of chemical shifts ppm spin spin line splitting H-1 phenol 1-H nmr explaining spin-spin coupling for line splitting doc brown's
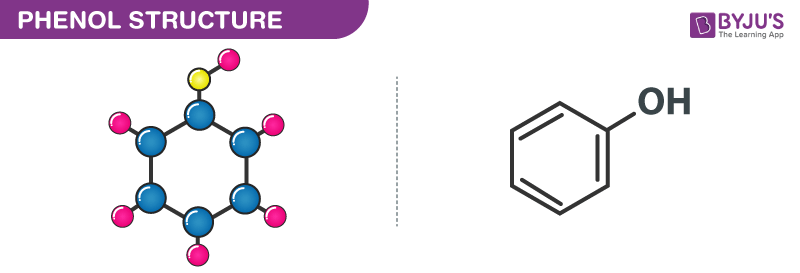
Phenol(carbolic acid)C6H5OH -Formula, Structure, Sources, Properties, and Uses of Phenol(carbolic acid)

Phenol (carbolic acid) — is an aromatic organic compound with the molecular formula C6H5OH. It is a white crystalline solid that is volatile Stock Photo - Alamy

Phenol(carbolic acid)C6H5OH -Formula, Structure, Sources, Properties, and Uses of Phenol(carbolic acid)

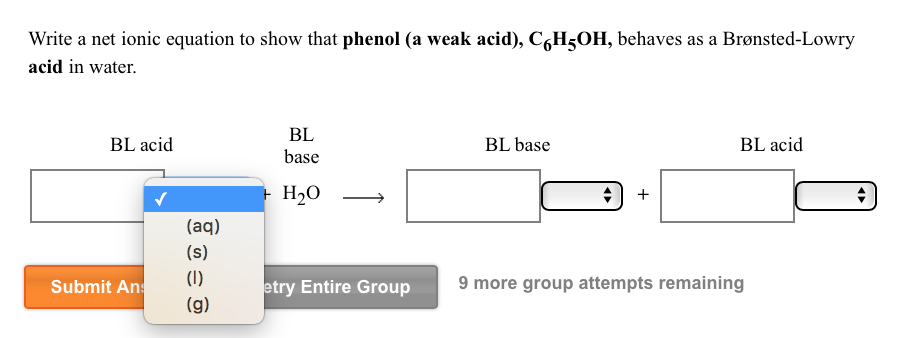
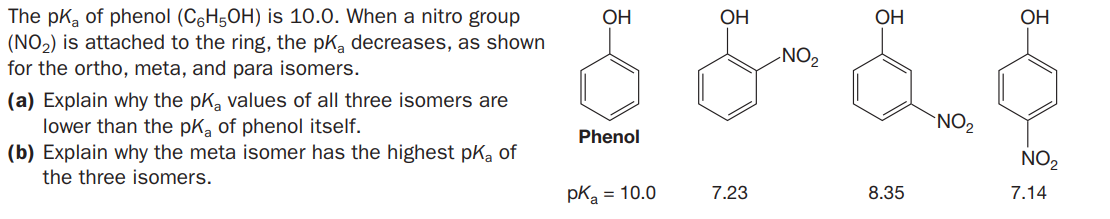
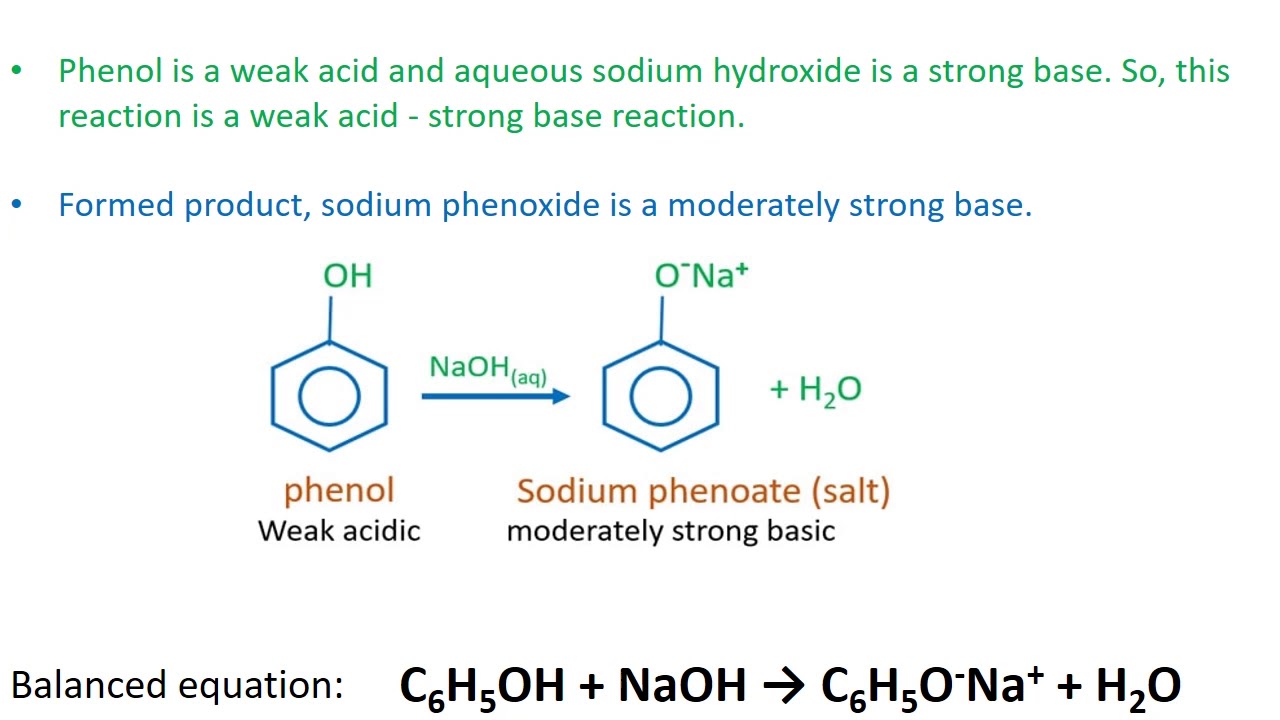
Which of the following bases are strong enough to deprotonate C6H5OH (pKa = 10) so that equilibrium favors the products? (a) H2O; (b) NaOH; (c) NaNH2; (d) CH3NH2; (e) NaHCO3; (f) NaSH; (