
First and second ionisation energy of Mg(g) are 720 k/J mol and 1440 kJ/mol respectively. Calculate the % of Mg^ + ions if one gram of Mg(s) absorbs 50 kJ of energy. (

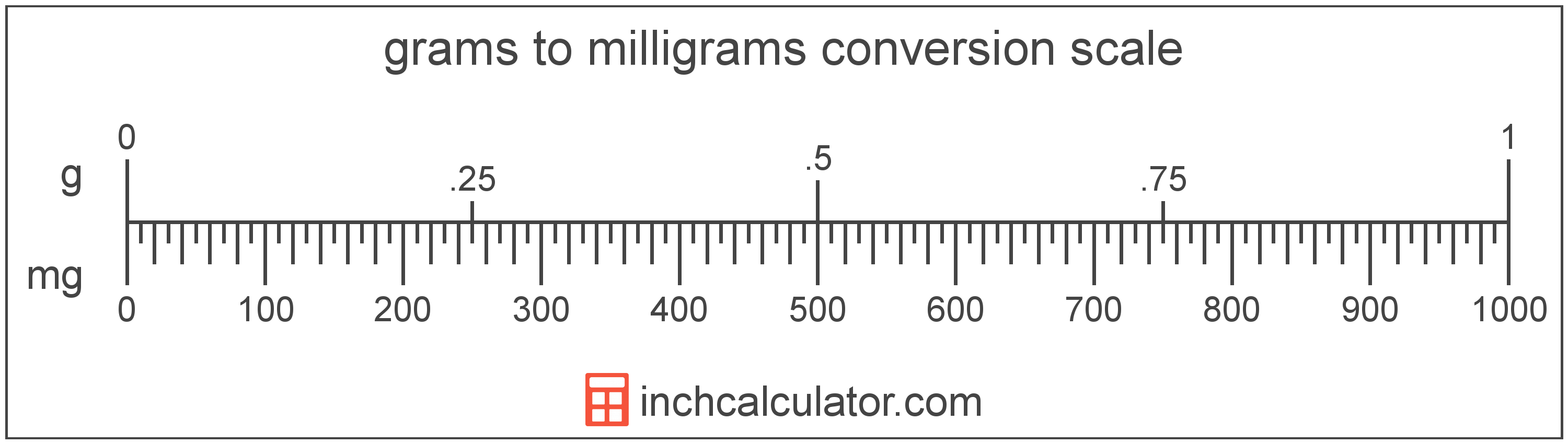
Ref: Gatford & Phillips (2002) Metric conversion 1 kilogram (kg) = 1000 grams (g) 1 gram (g) = 1000 milligrams (mg) 1 milligram (mg) = 1000 micrograms. - ppt download

Calculated Industries 8030 ConversionCalc Plus Ultimate Professional Conversion Calculator Tool for Health Care Workers, Scientists, Pharmacists, Nutritionists, Lab Techs, Engineers and Importers - Financial Calculators - Amazon.com

The number of molecules of methane ore present in 5.6 litres of methane at NTP is x × 10^23 . What is the value of x ?













/how-to-calculate-normality-609580final2-0d5efa5a961f4fa0a7efc780921faee1.png)