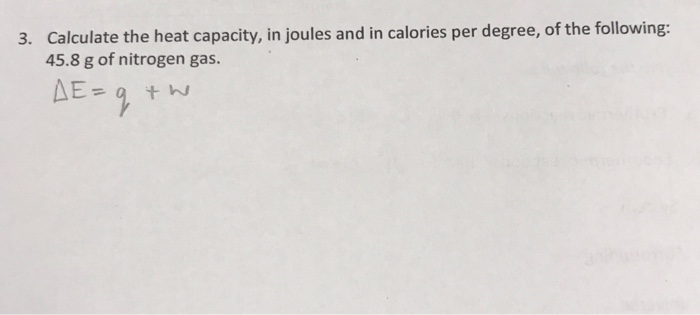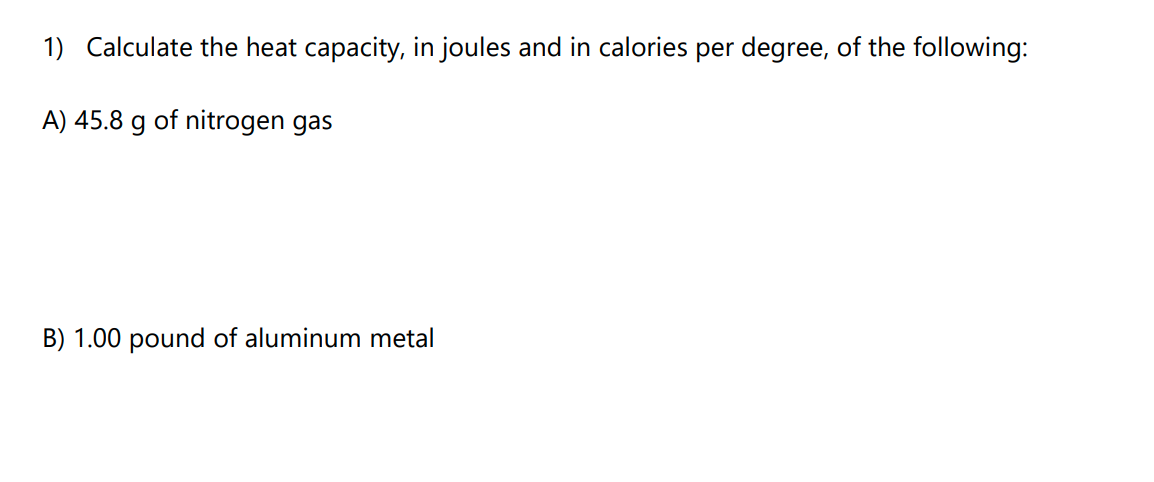The latent heat of fusion of ice is 80 "cal"//g at 0^(@)C what is the freezing point of a solution of Kcl in water containing 7.45 grm of solute in 500 grm

Worked example: Measuring the energy content of foods using soda-can calorimetry (video) | Khan Academy
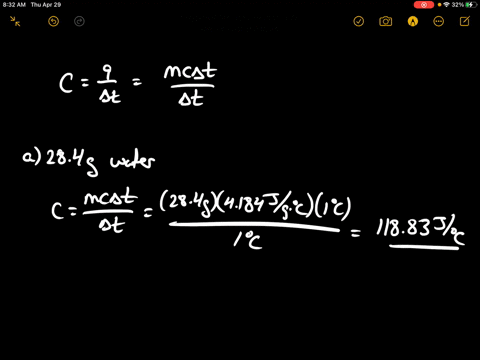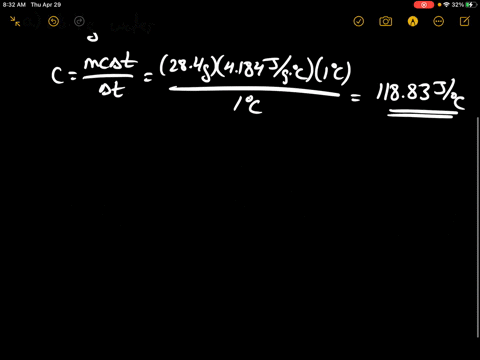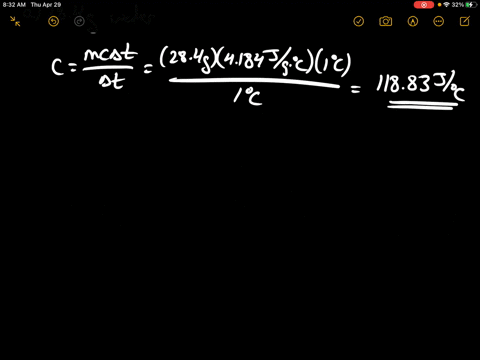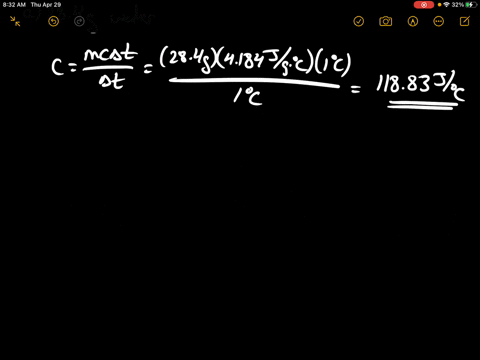
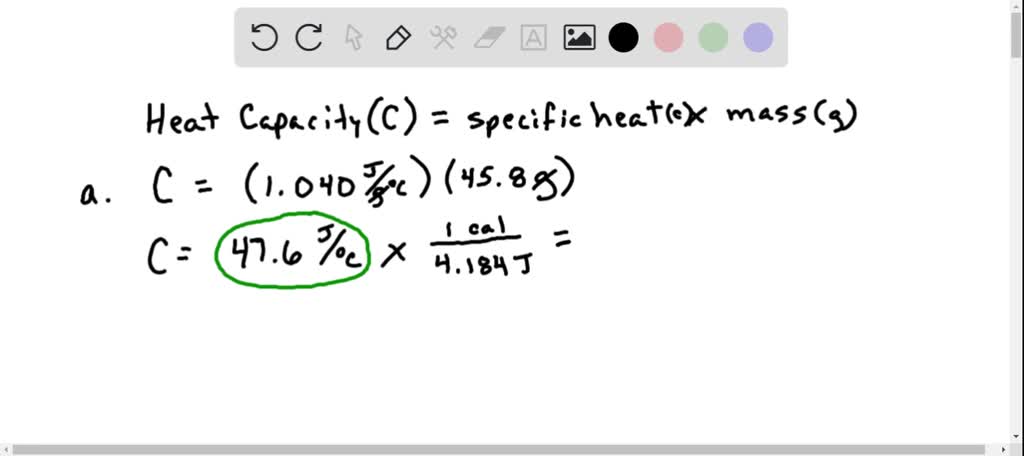
SOLVED:Calculate the heat capacity, in joules and in calories per degree, of the following: (a) 28.4 g of water (b) 1.00 oz of lead

SOLVED: Calculate the heat capacity, in joules and in calories per degree, of the following: (a) 28.4 g of water (b) 1.00 oz of lead

In a process, 10 gm of ice at - 5^oC is converted into steam at 100^oC .If latent heat of fusion of ice is 80 cal g^-1 , then the amount of
Additions and Corrections - The Rate of Reaction between Chlorate and Sulfur Dioxide in Acid Solution