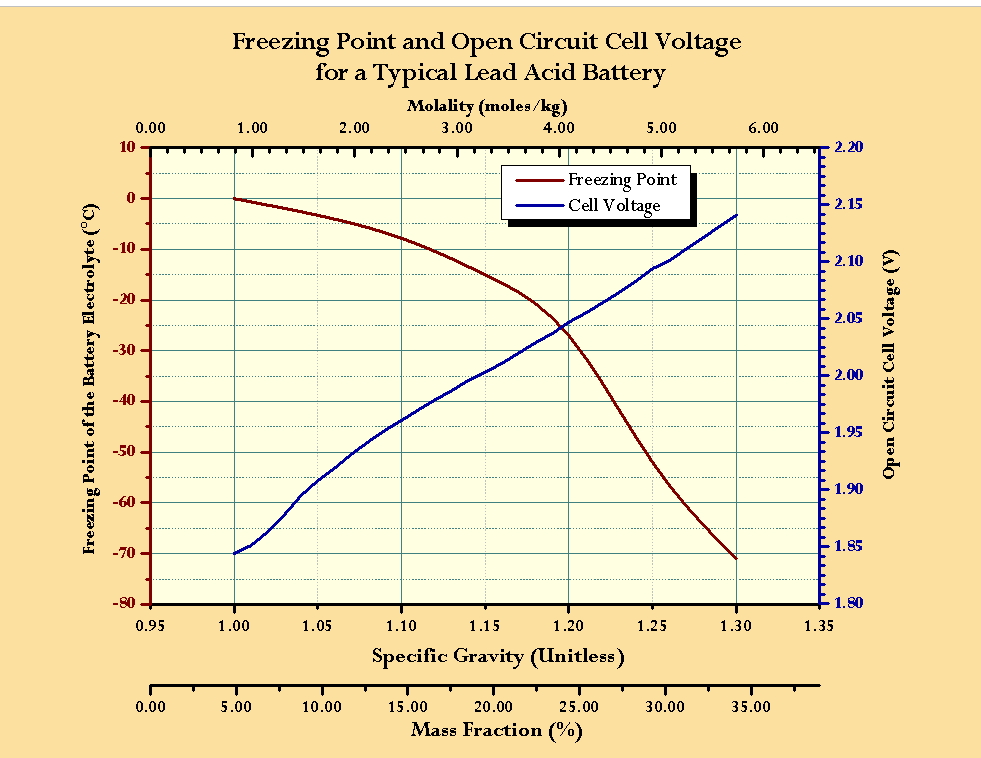
Battery acid is 4.27 molar h2 s o4 and has a density of 1.25 gram per ml what is the molality of h2s o4 in - Brainly.in

and 90 g of water have been mixed. A battery acid contains 24.5% by mass of H2SO4. What is the molality of the solution? A sample of sodium hydroxide weighing 0.48 g
Battery acid is 4.27M H2SO4(aq) and has density 1.25 g/ML. What is the molality of H2SO4 in the solution?

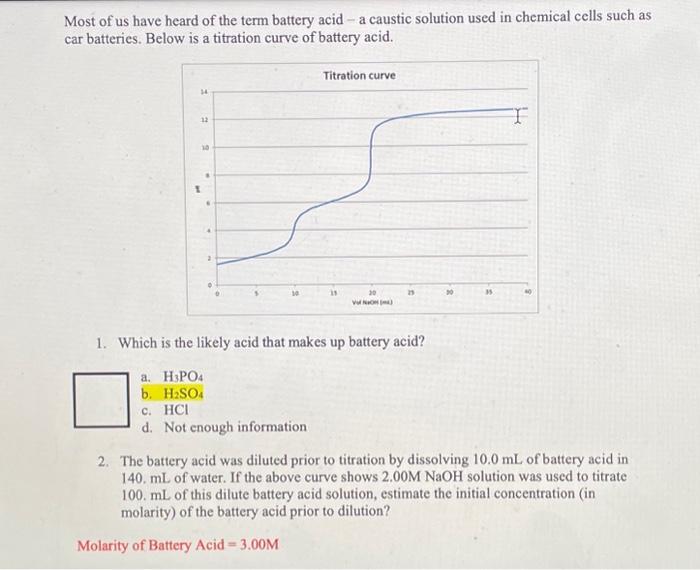
SOLVED: Called battery acid because it is used in car batteries, sulfuric acid is a very strong acid that can cause serious chemical burns upon contact. It can even lead to permanent

The molarity of sulfuric acid in a fully charged car battery is 5.2 m. when fully discharged the molarity - brainly.com

11. Battery acid is 4.27M H250, and has density of 1.25g/ml. What is the molality of HySO. In the solution?

please tell this question a battery acid contains 24 5% by mass of h2so4 what is the molarity - Chemistry - - 14507917 | Meritnation.com

11. Battery acid is 4.27M H250, and has density of 1.25g/ml. What is the molality of HySO. In the solution?








:max_bytes(150000):strip_icc()/car-battery-recycling-container-with-warning-notices-battery-acid-flusco-household-waste-recycling-centre-cumbria-uk-121814398-57a4e5055f9b58974a7355d8.jpg)