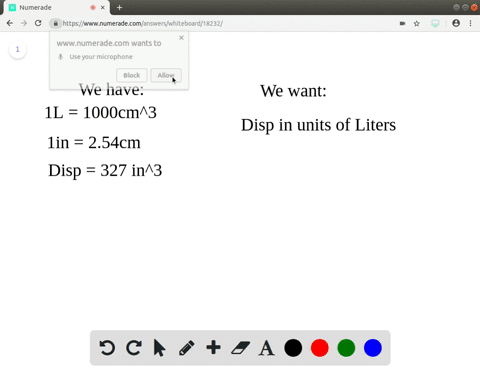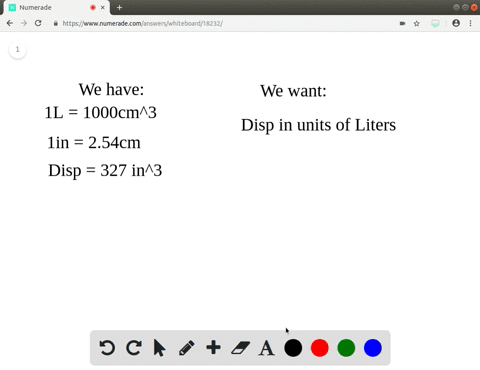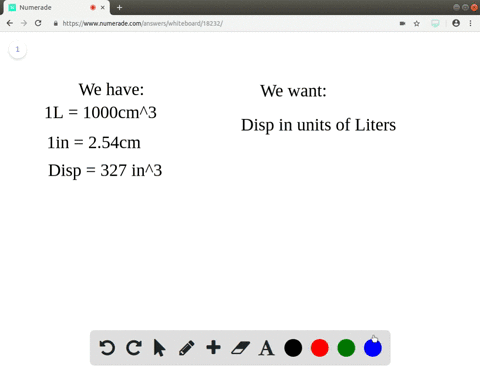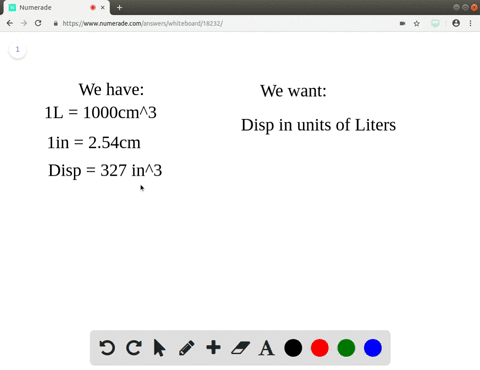
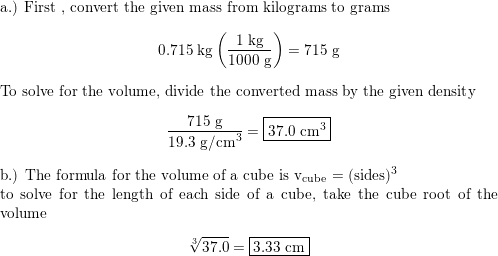A piece of pure gold of density `19.3 g cm^(-3)` is suspected to be hollow inside. It weighs 38.250 - YouTube

HW3_solutions - 4.4 Calculate the number of vacancies per cubic meter in gold (Au) at 900C. The energy for vacancy 3 formation is 0.98 eV/atom. | Course Hero

What kind of height and strength does a strong man need to hand over a tree? The mystery of MineCraft science – Hahapy

Gold crystallizes in a face centered cubic lattice. If the length of the edge of the unit cell is 407 pm, calculate the density of gold as well as its atomic radius

Density of gold is 19.30 g//cm^(3) at 20^(0)C. Computer the density of gold at 90^(0)C by adding steam to it. (alpha = 14.2 xx 10^(-6 //0) C)

SOLVED: Boltzmann constant; k = 8.62 X 10*eVlatom K, Avogadro $ constant;, NAv:= 6.022 x 1023 mol Calculate the equilibrium number of vacancies per cubic meter in gold (Au) at 258C For
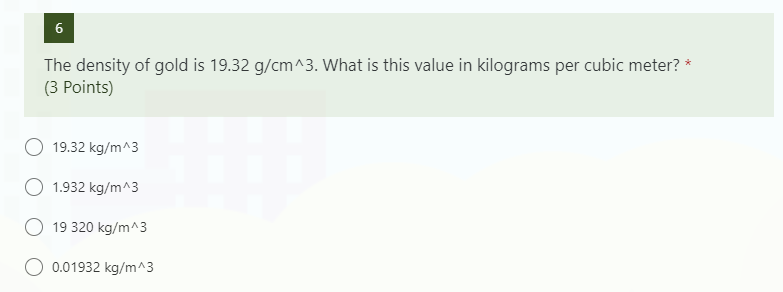
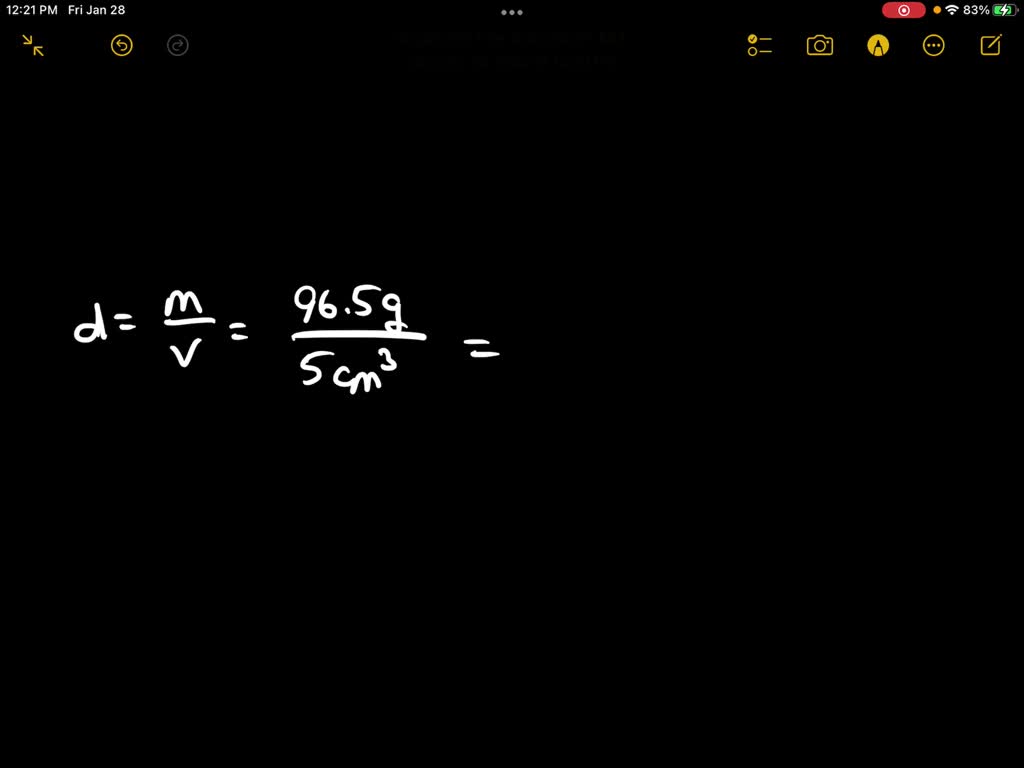
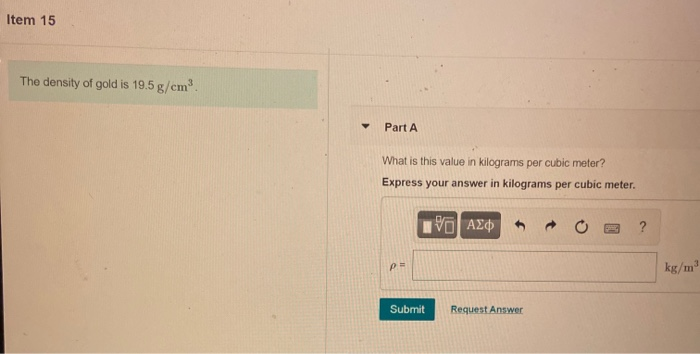
The density of gold is 19.300 kg/M3. What is the mass of a gold cube with a side length of 20.15 cm? - Quora

Gold has cubic crystals whose unic cell has edge length of 407.9 pm. Density of gold is 19.3 g cm^(-3). Calculate the number of atoms per unit cell. Also predict the type