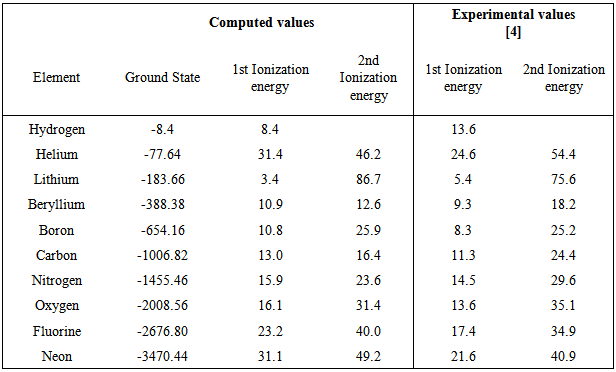
Computation of the First and Second Ionization Energies of the First Ten Elements of the Periodic Table Using a Modified Hartree-Fock Approximation Code
The second ionization energy is always greater than the first ionization energy. How is the second ionization of O more than N but the first ionization energy of N is more than

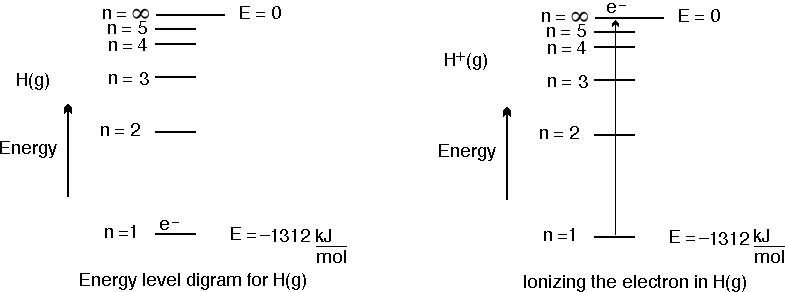
With the help of Bohr 's model , calculate the second ionisation energy of helium (energy required to remove the electron from He^(o+)


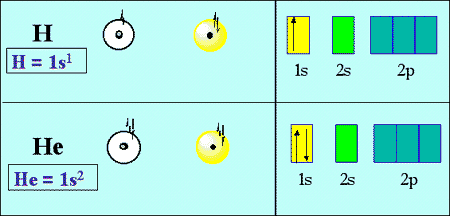
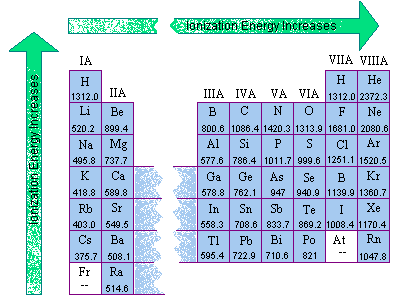




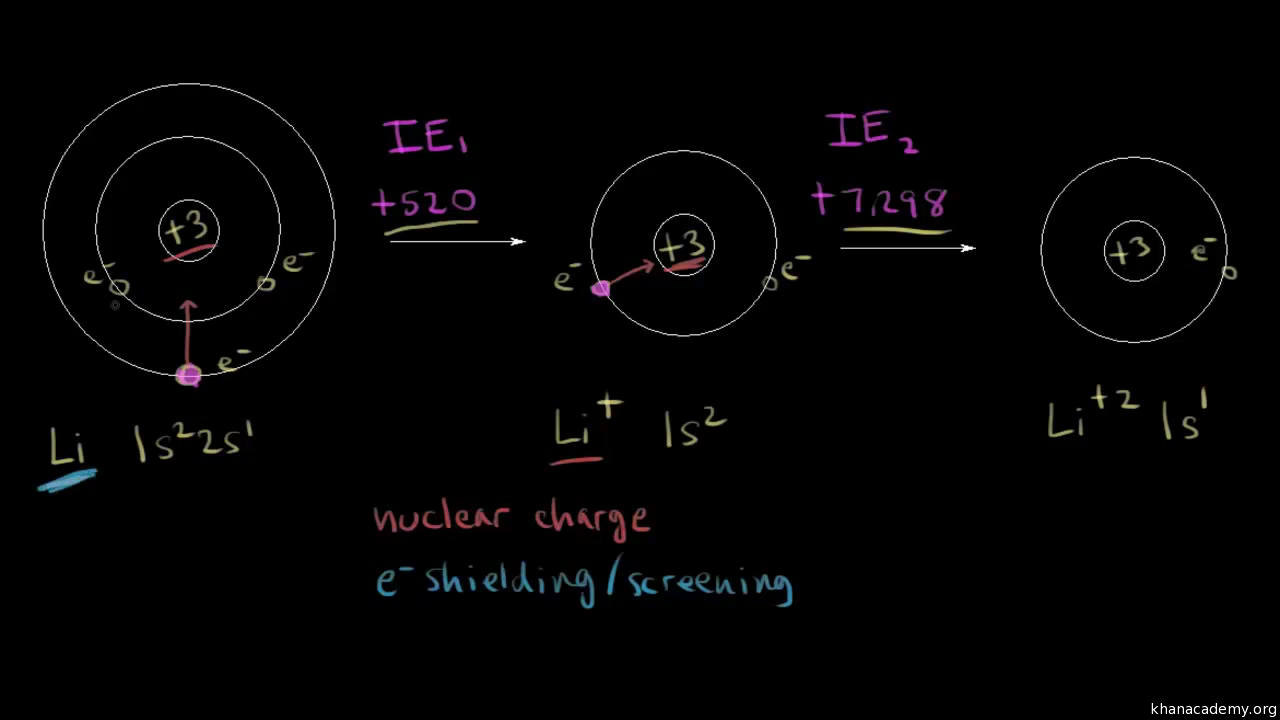
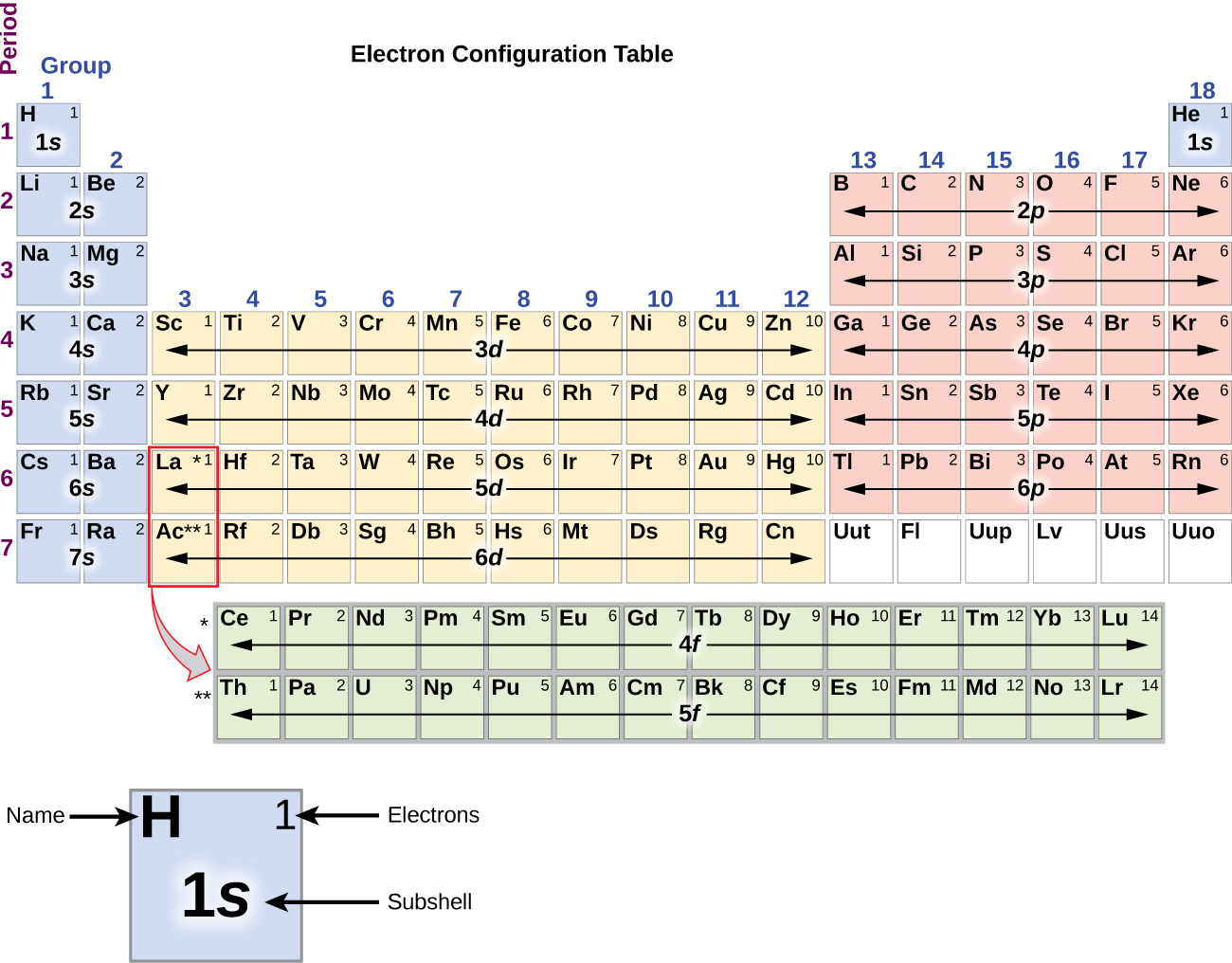
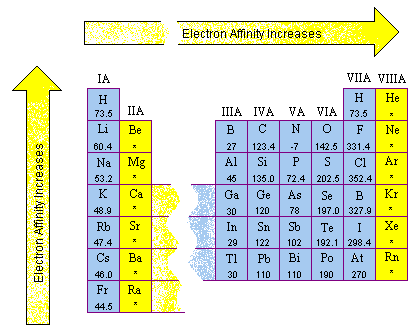
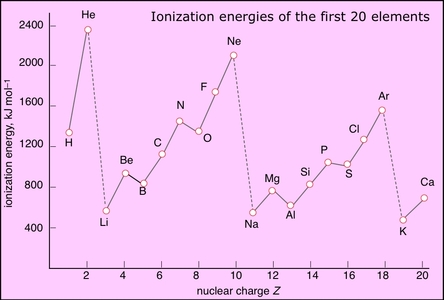

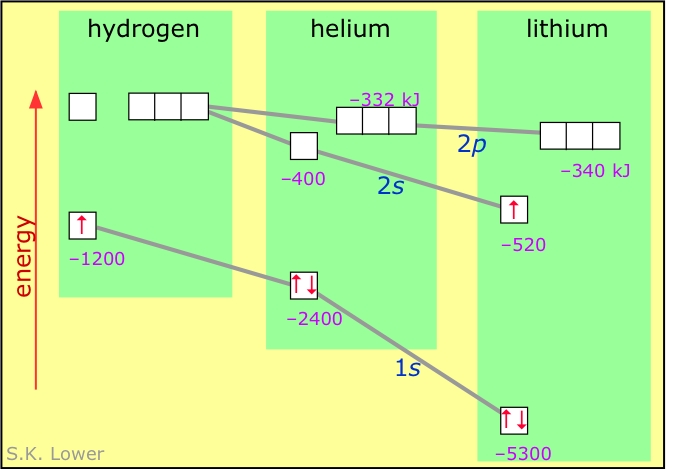
:max_bytes(150000):strip_icc()/800px-Orbital_representation_diagram.svg-589bd6285f9b58819cfd8460.png)