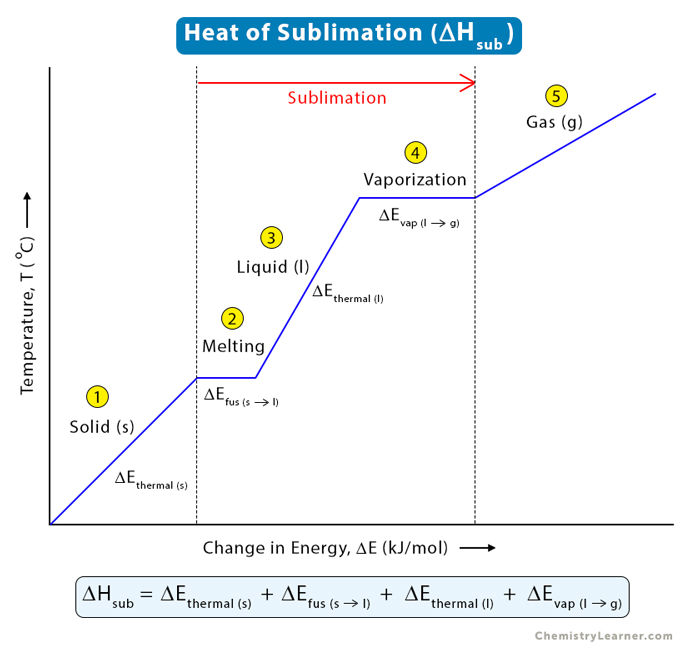
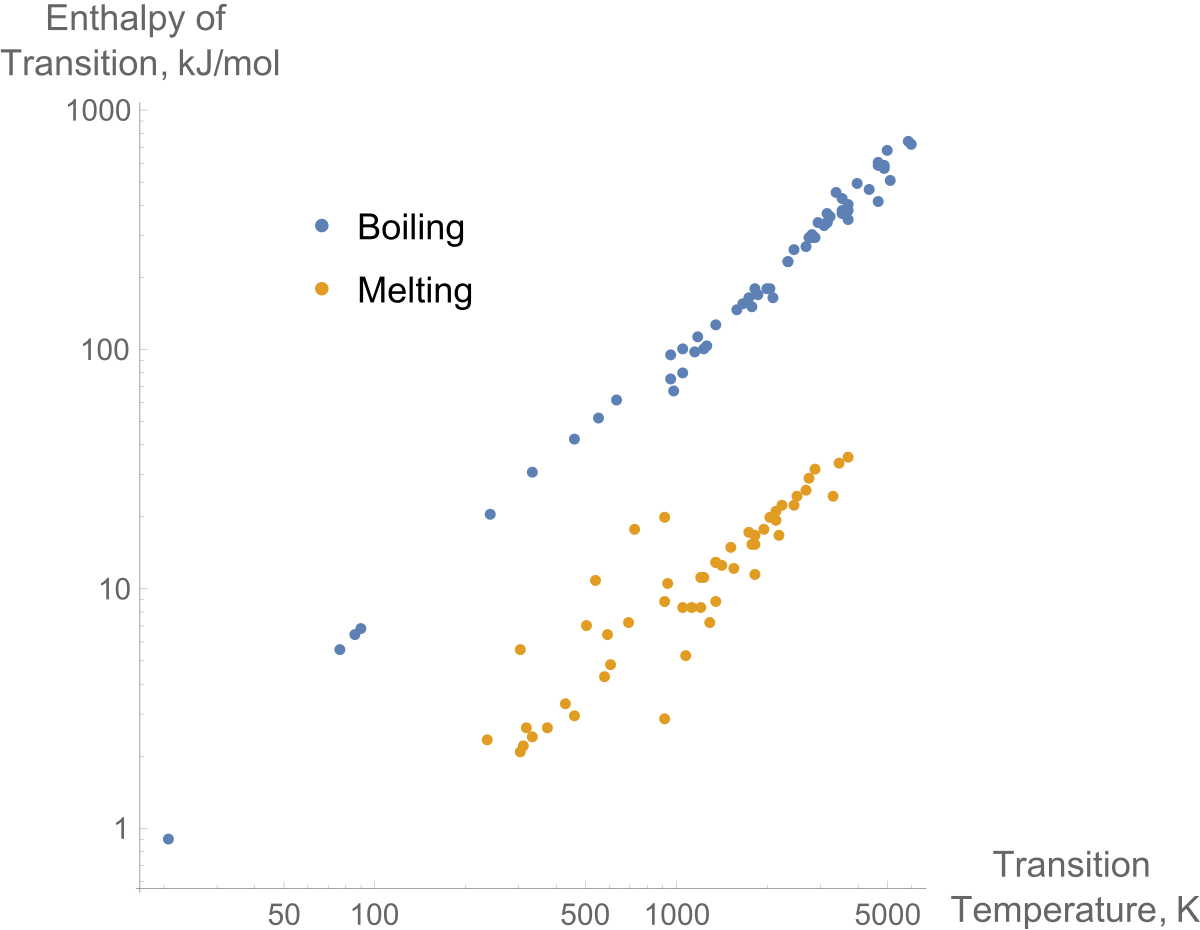
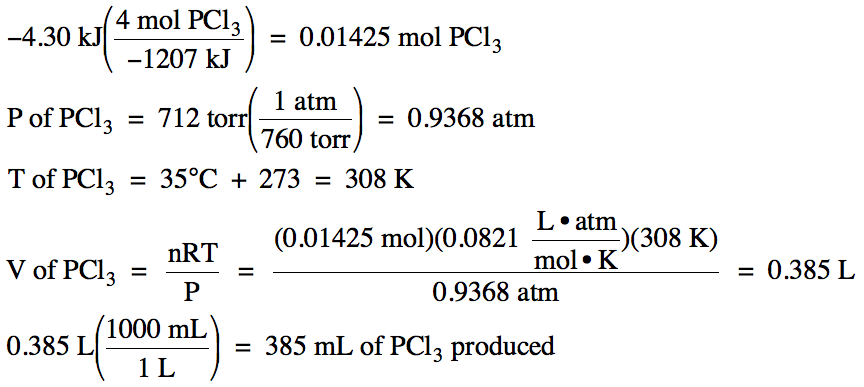
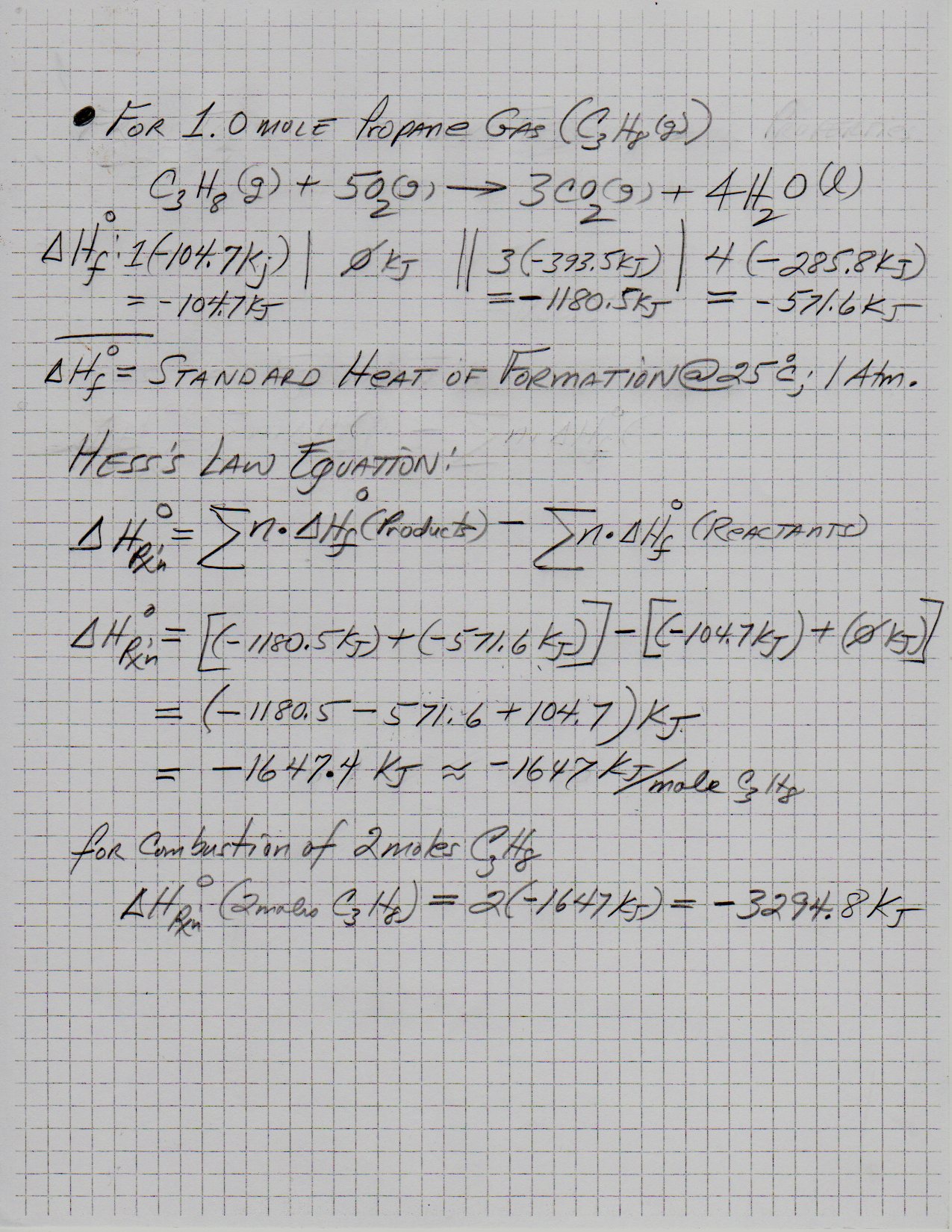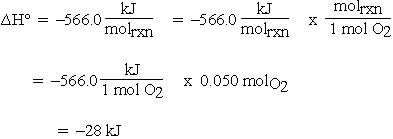Calculate the calorific value of methane if it burns according to the equation CH4(g) + 2O2(g) to CO2(g) + 2H2O(l) , DeltaH = -890.0 kJ

Homework 3 Solutions - Introductory Physical Chemistry | CH 331 | Assignments Physical Chemistry | Docsity

⚗️A student sets up the following equation to convert a measurement. (The stands for a number the - Brainly.com

SOLVED: The mass defect for an isotope was found to be 0.410 amulatom: Calculate the binding energy in kJ/mol of atoms: (1 J = kg m2/s2 ;lamu =1.66054e-24; 1 Mole equals 6.022x