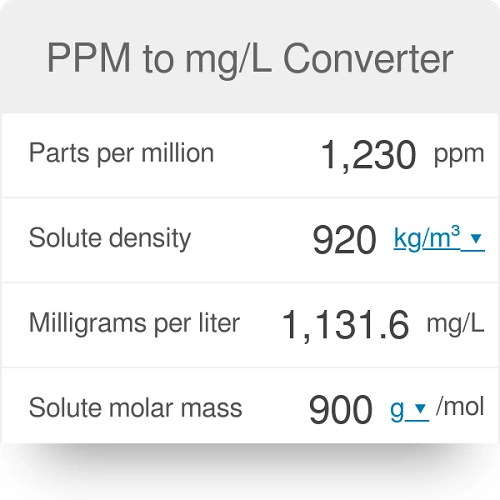
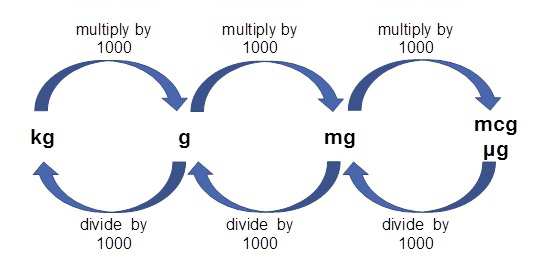
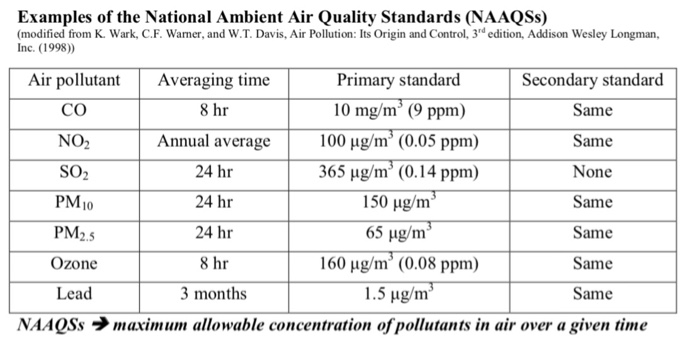
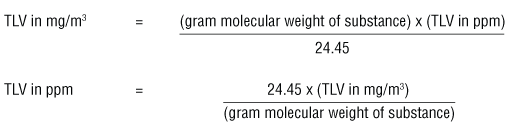SOLVED: 6.17 The dimensionless Henry's solubility constant (Cw/Ca) for ethanol is 4,885. In a laboratory the organic compound is measured at 1 ppm, which is 1,885 micrograms of ethanol per cubic meter
IAC Ch 26, p.1 567—26.2(455B) Episode criteria. 26.2(1) Evaluation. Conditions justifying the proclamation of an air poll